|
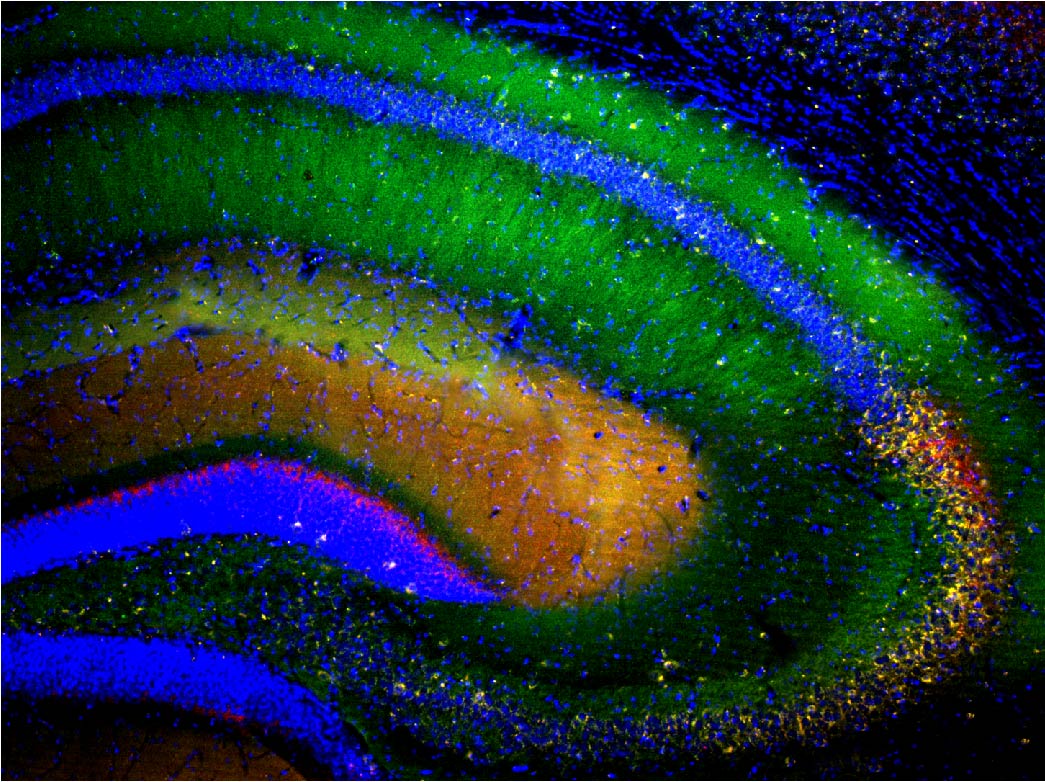
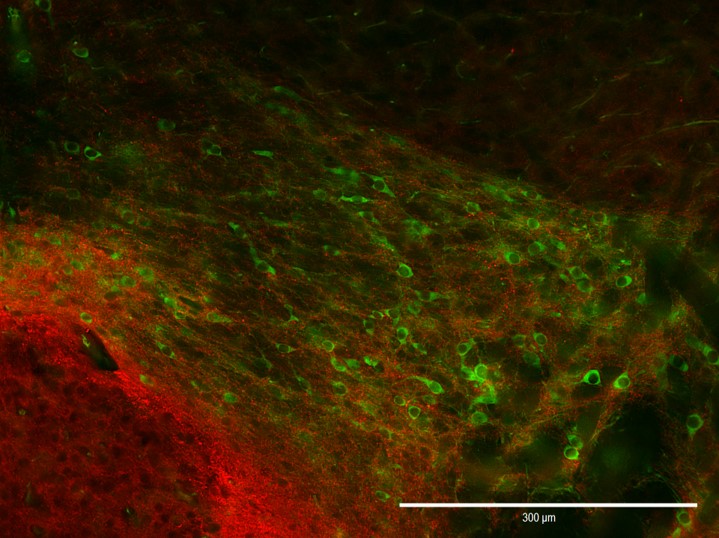
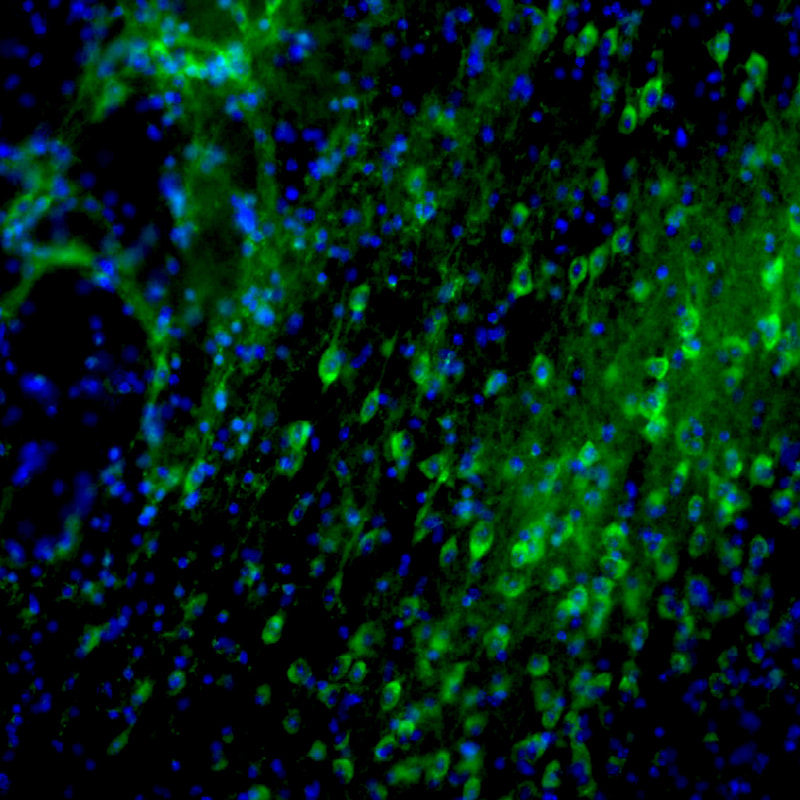
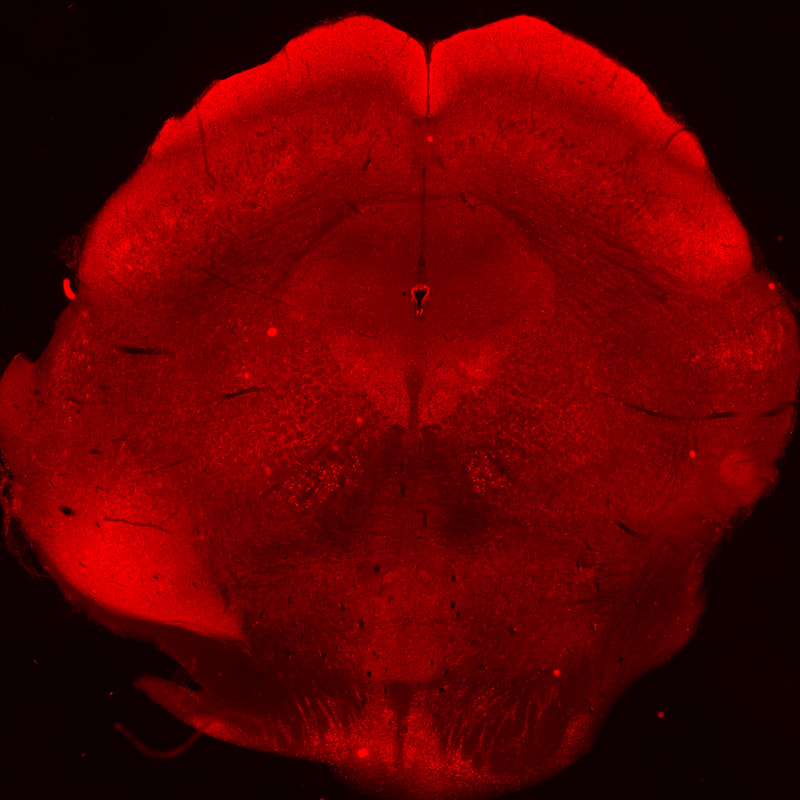
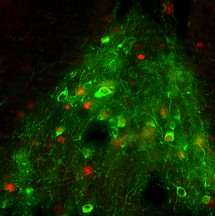
Cellular mechanisms of opioid tolerance and addiction Chronic pain is a huge problem that affects about one-third of the U.S. population. Despite many years of research for alternatives, opioids remain the most prescribed drugs to treat many types of pain, especially severe chronic pain, even though they possess many side effects including addiction. Novel therapeutics are needed to treat these patients. One area of focus in the lab is to determine the signaling mechanisms involved in opioid-induced analgesia that are altered, which leads to the development of tolerance to these drugs. When a person becomes tolerant to a drug, the dose must be increased in order to achieve the same level of pain relief, and that will lead to an increase in side effects. If we can determine what cellular changes cause tolerance, we can develop novel drugs that target these specific signaling pathways to better treat pain. Our previous studies reveled that commonly used opioids, such as morphine and fentanyl, produce analgesia using different signaling mechanisms, which leads to differences in the development of tolerance to these drugs. Ongoing studies in the lab will investigate blocking different intracellular molecules during long-term treatment with different opioids. In addition we are exploring the neuropeptide-receptor system (BigLEN-GPR171) in opioid functions. We find that GPR171 is highly expressed in areas involved in both addiction and pain and is found in cells that co-express mu opioid receptors. We found that following repeated opioid treatment there is an increase in GPR171 expression and signaling in various brain regions including the nucleus accumbens and amygdala. In addition, we see that blocking GPR171 function decreases morphine-induced reward, tolerance, and withdrawal. Future studies will determine which brain regions contribute to these behaviors. A novel neuropeptide receptor system, BigLEN-GPR171, in psychiatric disorders Psychiatric disorders including anxiety, depression, and substance use disorders, affect a large percentage of the population. Therapeutics to treat these patients are limited. A recently identified neuropeptide-receptor system (BigLEN-GPR171) shows high expression in many brain regions, such as basolateral amygdala, hippocampus, and prefrontal cortex, that contribute to stress- and fear-related behaviors that lead to these psychiatric disorders. Recently we have found that this system regulates body weight within the hypothalamus. Within the basolateral amygdala we find that this system regulates anxiety and fear, but not depression. Ongoing studies in the lab are investigating the cellular changes in BigLEN-GPR171 expression and signaling following stress or fear. |